Quantifying variation in labile traits
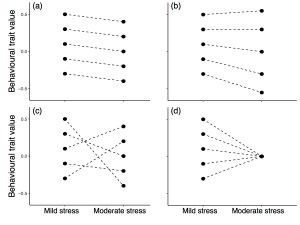
The majority of my postdoctoral research focuses on quantitative variation in traits that can change over time. One obvious example is behaviour, which we typically consider to be highly flexible such that individuals can change the way that they behave depending on the current environment. I use repeated observations on known individuals to determine what proportion of behavioural variation is due to differences among individuals (the ‘repeatability’ of the behaviour, which is often taken as a marker of ‘animal personality’). I am particularly interested in among-individual correlations between multiple behaviours (and whether these signal some underlying latent variable), and also between behaviours and other traits (such as physiological or neuroendocrine processes, or markers of individual fitness).
The repeatability of a labile trait can (under certain circumstances) provide an estimate of the upper limit of heritability, and I also use quantitative genetic methods to estimate how much of the phenotypic variation we see is due to additive genetic differences. Estimating genetic covariances between traits also helps us to understand how populations might (or might not) respond to selection.
In addition to the publications below, I have created various tutorials on using multivariate mixed modelling methods in the statistical software package R to partition (co)variation in repeated measures data.
Relevant publications:
T.M. Houslay, M. Vierbuchen, A.J. Grimmer, A.J. Young, A.J. Wilson (2017). Testing the stability of behavioural coping style across stress contexts in the Trinidadian guppy. Functional Ecology (in press). doi: 10.1111/1365-2435.12981
T.M. Houslay, A.J. Wilson (2017). Avoiding the misuse of BLUP in behavioural ecology. Behavioural Ecology, 28(4), 948-952. doi: 10.1093/beheco/arx023
D.K. Maskrey, A.J. Wilson, S.J. White, T.M. Houslay (2018). Who dares doesn’t always win: risk-averse rockpool prawns are better at controlling a limited resource. Animal Behaviour (in press).
Sexual selection and life history allocation

I am interested in sexual selection, especially regarding the evolution and maintenance of preference in species where there are no apparent direct benefits to the ‘choosy’ sex (typically females).
Variation in male fitness depends on differences in multiple traits, including exaggerated ornaments and weapons. When animals are competing for the attention of – or access to – potential mates, they often have at their disposal an incredible array of displays of sights, sounds and smells, not to mention fearsome weapons. How can we explain such diversity?
By integrating life history theory and sexual selection, we can compare the relative costs and benefits of these sexual traits. A universal cost involves the resources required to create and maintain them – if resources are used to make one trait, there is a cost incurred because those resources cannot be invested in any other traits. This ‘trade-off’ typically causes covariance between an organism’s resource budget and its level of sexual trait expression. We often talk about this in economic terms: the more money you have, then the more you have to spend (even after paying the bills!). This covariance between budget and sexual trait expression is known as condition-dependence.
Condition-dependence has several implications. It may be particularly important for ornaments signalling genetic quality, by helping to maintain the genetic variation that favours mate choice. It may also reduce extinction risk in sexually selected populations, by helping to weed out mutations that are detrimental to an individual’s overall vigour. Crickets make a useful study organism because the males have a ‘costly’ sexual trait – they rub their wings together to make a loud noise that attracts potential mates. Calling is costly because it requires energy to do it; the more energy a male has, the more calling he may be able to perform, and the more females he may attract. This type of trait means that different ‘strategies’ are available for the allocation of resources, as the investment level is highly dynamic. In my PhD, I used the decorated cricket Gryllodes sigillatus to study the interdependence of resource budgets and allocation strategies in a species where males express a sexually-selected trait repeatedly across their lifetime.
Relevant publications:
T.M. Houslay, K.F. Houslay, J. Rapkin, J. Hunt and L.F. Bussière. (2016), Mating opportunities and energetic constraints drive variation in age-dependent sexual signalling. Functional Ecology, 31, 728–741. doi:10.1111/1365-2435.12766
T.M. Houslay, J. Hunt, M.C. Tinsley, and L.F. Bussière. (2015), Sex differences in the effects of juvenile and adult diet on age-dependent reproductive effort. Journal of Evolutionary Biology, 28, 1067–1079. doi: 10.1111/jeb.12630
T.M. Houslay. and L.F. Bussière. (2012), Sexual selection and life history allocation. Encyclopaedia of Life Sciences.